Consequently, there is no secondary (welfare) standard for CO.ĬO contributes indirectly to climate change because it participates in chemical reactions in the atmosphere that produce ozone, which is a climate change gas. EPA conducted an extensive literature search as part of the review of the National Ambient Air Quality Standard for Carbon Monoxide that was completed in 2011, and did not identify any evidence for ecological effects of CO at levels at or near ambient. How does carbon monoxide affect the environment? 
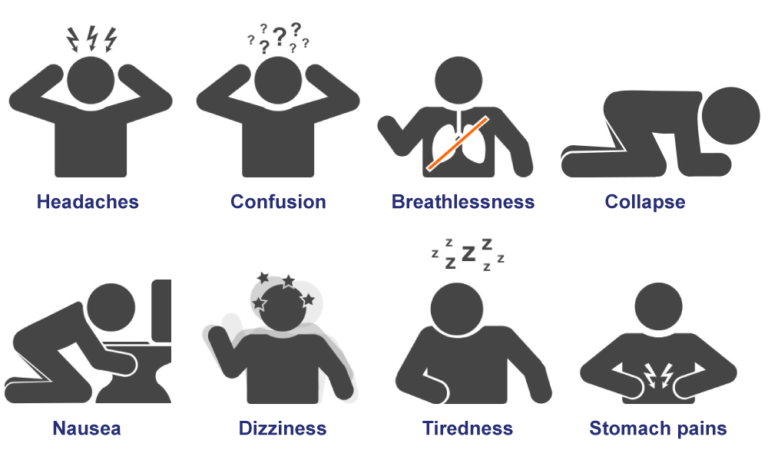
Unborn babies, infants, elderly people, and people with anemia or with a history of heart or respiratory disease are most likely to experience health effects with exposure to elevated levels of CO. Who is at the greatest risk from exposure to carbon monoxide? Unborn babies whose mothers experience high levels of CO exposure during pregnancy are at risk of adverse developmental effects. Inadequate oxygen delivery to the heart muscle leads to chest pain and decreased exercise tolerance. For people with cardiovascular disease, short-term CO exposure can further reduce their body’s already compromised ability to respond to the increased oxygen demands of exercise, exertion, or stress. The most common effects of CO exposure are fatigue, headaches, confusion, and dizziness due to inadequate oxygen delivery to the brain. This interferes with oxygen delivery to the body’s organs. What kinds of harmful effects can carbon monoxide cause?Ĭarbon monoxide is harmful because it binds to hemoglobin in the blood, reducing the ability of blood to carry oxygen. 
There is substantial evidence that CO can adversely affect health, participate in atmospheric chemical reactions that result in formation of ozone air pollution, and contribute to climate change. EPA focus on carbon monoxide?Īir quality regulators are concerned about air pollutants which may reasonably be anticipated to endanger public health and welfare. There are also a number of indoor sources of CO that contribute to total exposure. Carbon monoxide can also be formed through photochemical reactions in the atmosphere from methane and non-methane hydrocarbons, other volatile organic hydrocarbons in the atmosphere, and organic molecules in surface waters and soils. Nationally and, particularly in urban areas, the majority of outdoor CO emissions to ambient air come from mobile sources. It results from the incomplete combustion of carbon-containing fuels such as natural gas, gasoline, or wood, and is emitted by a wide variety of combustion sources, including motor vehicles, power plants, wildfires, and incinerators. Carbon monoxide (CO) is a colorless, odorless gas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
